IPLab:Lab 13:Wilms Tumor
Contents
Clinical Summary[edit]
The mother of this three-year-old male noticed a large mass in his abdomen. There were no other complaints. Physical examination and x-rays--including an intravenous pyelogram (IVP)--showed a large right renal mass. At surgery, a 550-gram right kidney was removed. There was no lymph node involvement, no extrarenal mass, and no involvement of the other kidney. Lung and bone x-rays showed no metastases. However, microscopic study of the surgical specimen confirmed a diagnosis of Wilms tumor and showed infiltration by the tumor (Grade II neoplasm) which had been difficult to separate from the duodenum. The child was placed on chemotherapy. Four months after the surgery to remove the tumor, a nodule was found in the lungs. The chemotherapy was resumed but the patient died four weeks later due to complications of Gram-negative sepsis.
Images[edit]
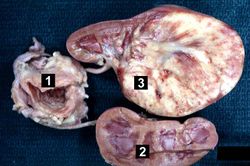
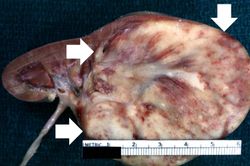
This autopsy specimen if from a similar case of a child who died of Wilms tumor. This gross photograph shows the bladder (1) to which are attached a normal kidney (2) and a kidney with Wilms tumor (3). A large mass extends from the superior pole of the affected kidney. The renal capsule can be seen extending around this tumor.
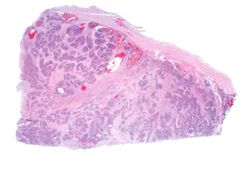
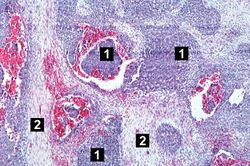
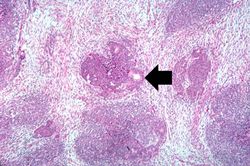
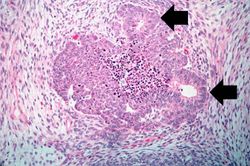
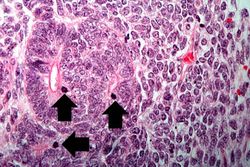
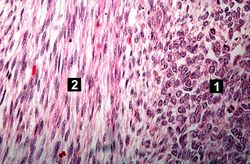
This lowest-power view shows the tumor itself; no tissue is present that can be readily identified as normal kidney. There does appear to be a capsule surrounding the tumor. Eosinophilic bands are seen surrounding basophilic islands of cells. These correspond to the two types of tissue in this tumor--the basophilic cellular component termed "blastema" can be distinguished from less cellular eosinophilic areas.
Virtual Microscopy[edit]
Study Questions[edit]
Additional Resources[edit]
Reference[edit]
- eMedicine Medical Library: Surgery for Wilms Tumor
- eMedicine Medical Library: Wilms Tumor
- Merck Manual: Wilms' Tumor
Journal Articles[edit]
- Vadeyar S, Ramsay M, James D, O'Neill D. Prenatal diagnosis of congenital Wilms' tumor (nephroblastoma) presenting as fetal hydrops. Ultrasound Obstet Gynecol 2000 Jul;16(1):80-3.
Images[edit]
| |||||
The patient is given an intravenous injection of contrast medium that rapidly enters the urine. Radiographs are then taken to show the passage of the contrast-containing urine through the pelvicaliceal system.
A normal kidney for a child this age should weigh 50 grams.
Gram-negative sepsis is a systemic inflammatory response caused by the presence of Gram-negative bacteria and their endotoxins in the blood. The microbes usually enter the bloodstream from an area of localized infection. Gram-negative sepsis is the leading cause of septic shock--hence, also known as "endotoxic shock." Septic shock is a syndrome of inflammatory responses which can lead to severe hypotension, multiple organ system failure, and death.
Renal failure is the severe reduction of renal function and often leads to reduced urinary output.
The normal fibrinogen level is 184 to 412 mg/dL.