IPLab:Lab 9:Diphtheria
Contents
Clinical Summary[edit]
This 4-year-old black female had an upper respiratory infection and a sore throat with increasing difficulty in breathing. Membranous exudate over one tonsil led to a working diagnosis of diphtheria, and the child was admitted. On the day of her admission, the child developed signs of respiratory tract obstruction and a tracheotomy was performed. However, the procedure was unable to establish a patent airway and the child died.
At autopsy, a dense grayish pink membrane extended from both tonsils to the mid-trachea. The lungs were edematous and showed signs of pneumonia.
Images[edit]
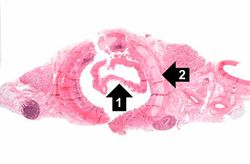
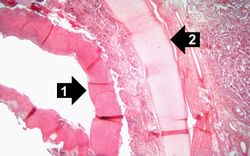
This is a higher-power photomicrograph of trachea with the diphtheritic membrane (1). Even though, the main part of the membrane has pulled away from the tracheal lining during histological processing, in this section part of the membrane is still loosely attached. Once again, note the tracheal cartilage (2).
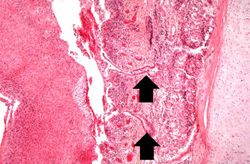
This is an even higher-power photomicrograph of the tracheal mucosa and the diphtheritic membrane. The mucosal surface of the trachea is ulcerated (total loss of epithelial cells) and the only remaining epithelial cells are found in the glands (arrows). The diphtheritic membrane consists of fibrin and inflammatory cells, most of which are dead.
Virtual Microscopy[edit]
Study Questions[edit]
Additional Resources[edit]
Reference[edit]
- eMedicine Medical Library: Diphtheria
- eMedicine Medical Library: Pediatric Pneumonia
- eMedicine Medical Library: Corynebacterium Infections
- Merck Manual: Diphtheria
Journal Articles[edit]
- Zakikhany K, Efstratiou A. Diphtheria in Europe: current problems and new challenges. Future Microbiol 2012 May;7(5):595-607.
Images[edit]
| |||||
In alcoholics, aspiration pneumonia is common--bacteria enter the lung via aspiration of gastric contents.